|
Classification of Amines, Electrophilic Aromatic Substitution in Aromatic. F) the substitution follow a stepwise mechanism. The overall electrophilic aromatic substitutions are exergonic reactions. Position in the Periodic Table of Transition and Inner Transition Elements. reactivity of aromatic substitution (both electrophilic and nucleophilic). In the table below, you can see that some substituents confer a rate of reaction that is much higher than. It has a lower free energy of activation. electrophilic aromatic substitution reactions. The second step is fast and exergonic because it restores the stability enhancing aromaticity. This step is therefore the rate-determining step because of its higher free energy of activation. The free energy diagram shows that the first step is relatively slow and endergonic because the ring loses its aromatic stability. In the second step, deprotonation of the arenium ion restores aromaticity and gives the substituted product. 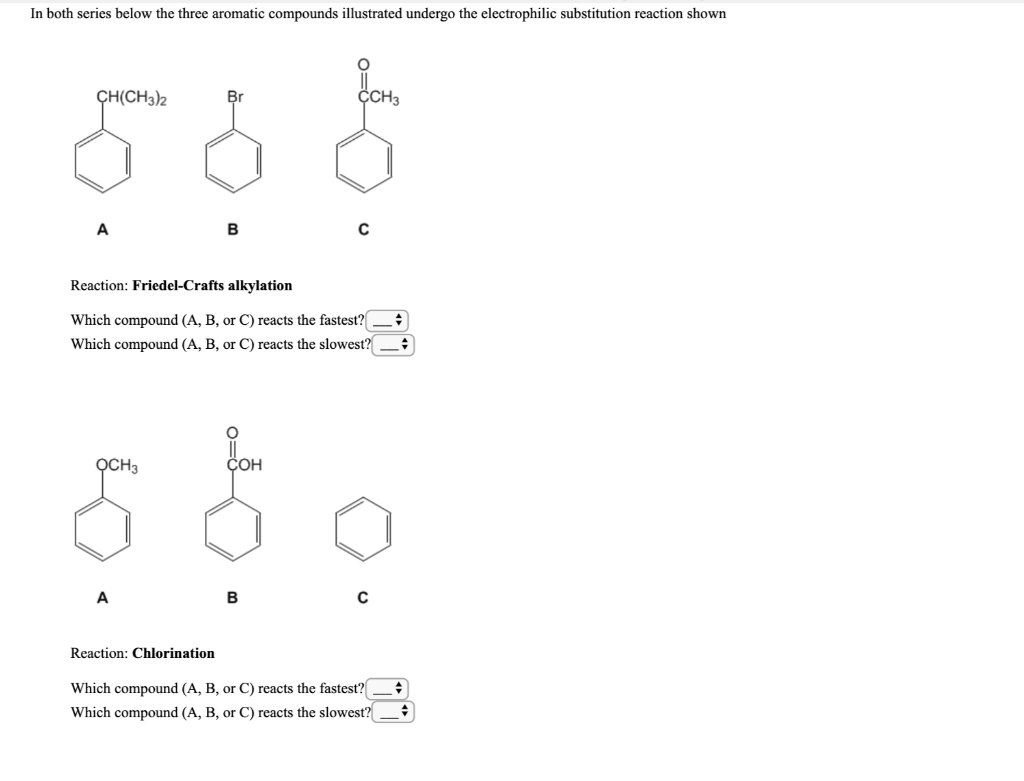
It is often referred to as a sigma complex because the electrophile forms a sigma bond with the aromatic ring. In the first step, the π system of the aromatic ring reacts with an electrophile, forming an arenium ion, which is resonance-stabilized. All electrophilic aromatic substitution reactions occur via a two-step mechanism. Many functional groups can be added to aromatic compounds by these reactions. In an electrophilic aromatic substitution reaction, an electrophile substitutes for a hydrogen of an aromatic compound. Overall, electrophilic aromatic substitutions are exergonic reactions. It has lower free energy of activation and is fast. In contrast, the second step is exergonic because it restores aromatic stability to the system. It is, therefore, the rate-determining step. This step has a higher free energy of activation and is slow. In the second step, the arenium ion is deprotonated, restoring aromaticity and giving the substituted product.Īs evident from the free energy diagram, the first step is endergonic because the ring loses its aromatic stability. The arenium ion is also called a sigma complex because the electrophile forms a sigma bond with the aromatic ring. Table 3-2: MPEX, Harel and Hammer Analogies. In the first step of the reaction mechanism, the π system of the aromatic ring attacks the electrophile to form an arenium ion, which is resonance stabilized. Arenes contain double bonds just like alkenes but they do not undergo electrophilic addition because these would result to their loss of ring aromaticity. 
These reactions allow the introduction of different functional groups onto aromatic rings. Aromatic compounds or arenes undergo substitution reactions, in which the aromatic hydrogen is replaced with an electrophile, hence their reactions proceed via electrophilic substitution. Electrophilic aromatic substitutions are reactions in which an electrophile replaces one of the aromatic hydrogens.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
